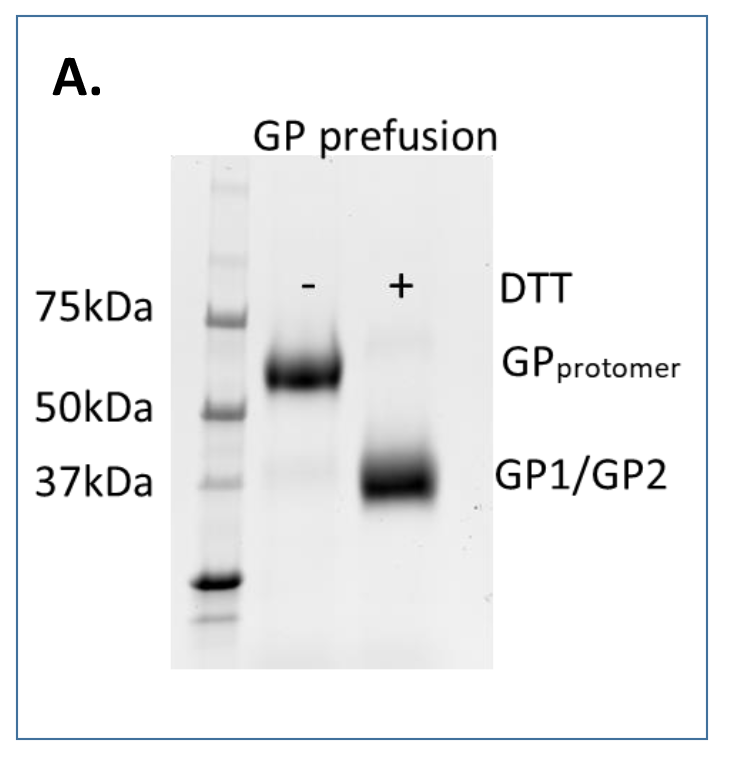
Glycoproteins secreted into the supernatant of Drosophila S2 lines are purified via streptactin affinity. Following elution proteins are dialyzed into PBS, filtered, aliquoted and flash frozen in LN2. Non-reducing and reducing SDS-PAGE gels are run to analyze purity from strep-affinity purification and, for GPCysR4 proteins, efficient furin-mediated processing (A.).
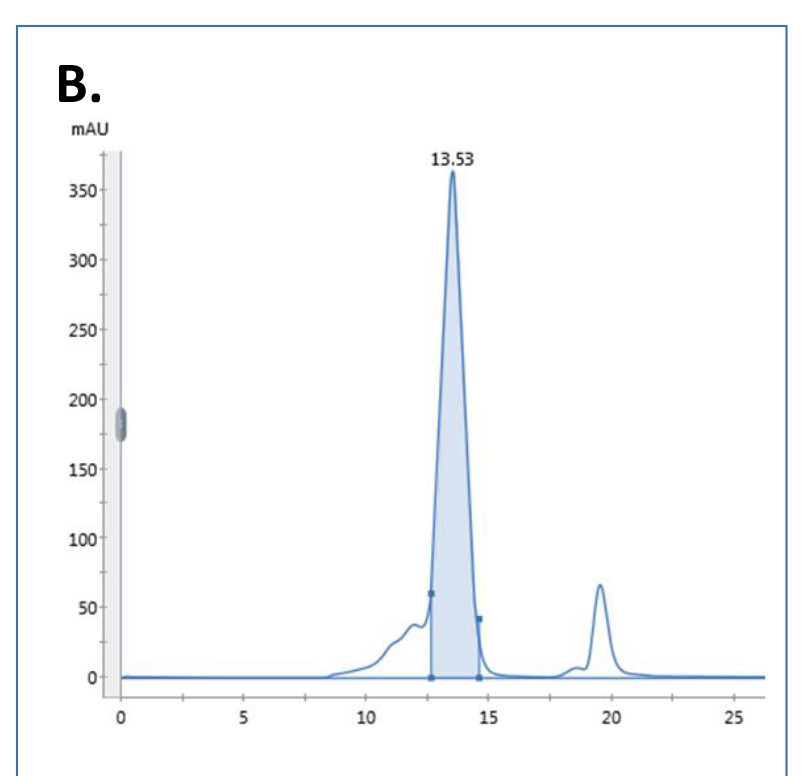
New constructs are analyzed by SEC-MALS to determine molecular weight, oligomeric state and glycan content (B.).
For each new lot of GP, an ELISA screen is performed with a variety of antibodies to ensure the same antibody reactivity profile is consistent (C.). Antibodies used in ELISA screens include: 2.4F and 5.6H – GP1, non-neut; 12.1F and 19.7E – GP1, neut; binds upper portion of GP1 subunit. NB: 19.7E is not cross reactive to Lineage III; 3.6H and 13.9H – GP2, post fusion specific, non-neut; 18.5C – GPC-B, neut; quaternary – binds at the base of GP and simultaneously contacts two GP monomers; 22.5D – linear GP2, pan arena; 25.10C and 36.1F – GPC-A – neut, quaternary epitope; binds mid-upper portion of GP, contacts both GP1 and GP2 subunits from the same monomer, does not bind across trimers. NB: 36.1F is Lineage IV specific in neutralization studies.
Lassa Glycoproteins, Viral Antigens
(LASV-GP-014) LASV Glycoprotein antigen, Strain Pinneo, Ectodomain, Pre-fusion GP monomer, lineage I
$1,680.00 – $3,350.00
Designation: LASV GPC, Pinneo, ectodomain, pre-fusion heterodimeric GP1-GP2 complex
Clade specificity: I
Produced in: S2 Drosophila cells
Applications: ELISA, WB, antigen capture, structural studies
Format: liquid, frozen
Formulation: 1xPBS, pH 7.4
Storage: -80°C, in aliquots
Shipping: frozen, dry ice
SKU: LASV-GP-014
Categories: Lassa Glycoproteins, Viral Antigens
Related products
-
Lassa Nucleoproteins
(LASV-R-0032) LASV NP, N-term, 1-340 lineage III
$600.00 – $1,825.00 Select options This product has multiple variants. The options may be chosen on the product page -
Lassa Nucleoproteins
(LASV-R-0021) LASV NP, full length lineage II
$600.00 – $1,825.00 Select options This product has multiple variants. The options may be chosen on the product page -
Lassa Glycoproteins
(LASV-GP-010) LASV Glycoprotein antigen, Strain Pinneo, Ectodomain, Linked, lineage I
$1,680.00 – $3,350.00 Select options This product has multiple variants. The options may be chosen on the product page -
Lassa Nucleoproteins
(LASV-R-0062) LASV NP, C-term, Δ 340 lineage VI
$600.00 – $1,825.00 Select options This product has multiple variants. The options may be chosen on the product page