This assay is a direct ELISA detecting Human IgG antibodies specific for SARS CoV-2 nucleoprotein (N Protein). Diluted samples, Reference, Positive and Negative Controls are incubated in microwells coated with a mixture of recombinant SARS CoV-2 N antigens. Incubation allows the anti-SARS CoV-2 antibody present in the samples to react with the immobilized antigen mixture. After the removal of unbound serum or plasma proteins by washing, anti-human IgG antibodies, labeled with horseradish peroxidase (HRP), are added forming complexes with the bound IgG anti-SARS CoV-2 antibody. Following another washing step, the bound enzyme-antibody conjugate is assayed by the addition of TMB substrate. Color develops in the wells at an intensity proportional to the concentration of IgG anti-SARS CoV-2 antibody in the sample. Optical Density (O.D.) results are obtained by reading the absorbance (A450nm minus A620nm) using an ELISA plate reader. It is recommended that the user establish a cut-off for the study population using SARS CoV-2 sero-negative samples. It is also recommended that IgG positive convalescent SARS CoV-2 samples from the study population be included in each assay as an additional reference sample.
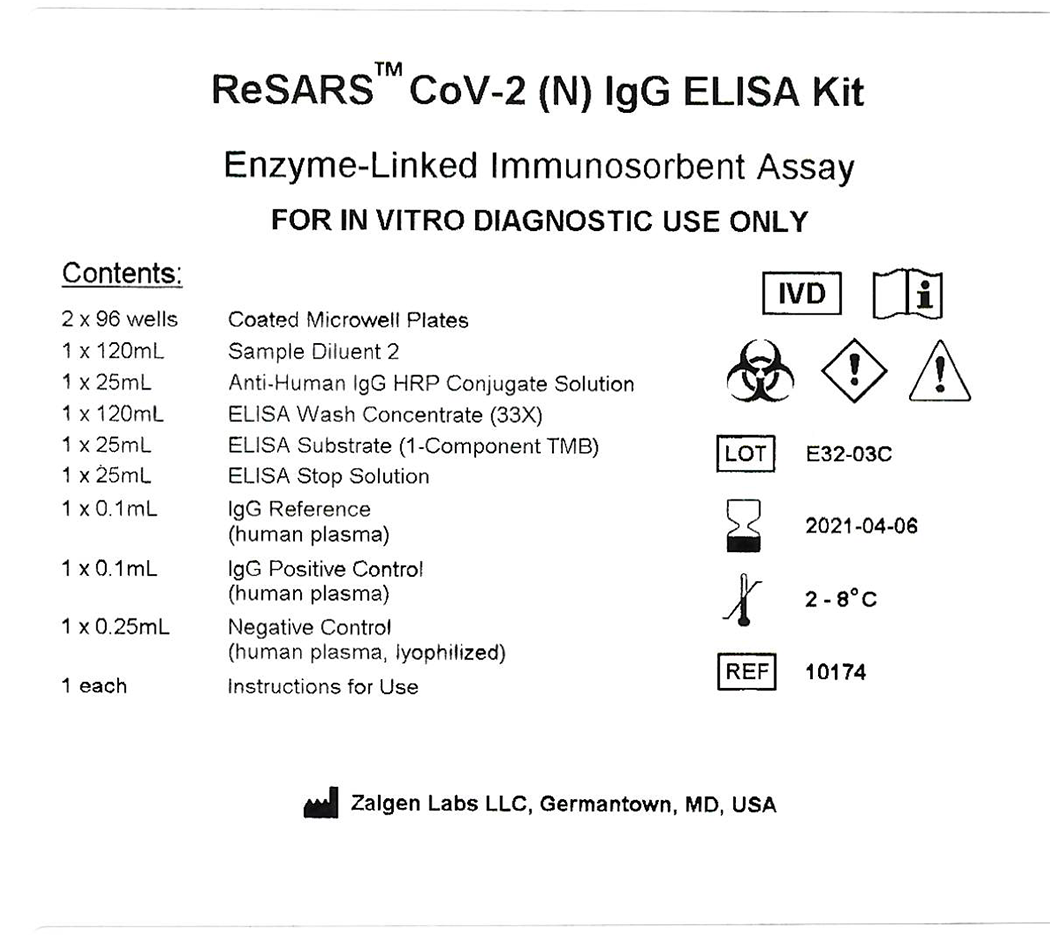
Package insert for 10174 ReSARS CoV-2 (N) IgG ELISA (IVD) pdf